
From screening to triage: how quantitative FIT optimises colorectal cancer diagnostics
FIT testing in colorectal cancer screening
Colorectal cancer (CRC) is a significant global health challenge. It is the third most common cancer and the second most common cancer-related cause of death worldwide. Around 1.9 million colorectal cancer cases are diagnosed annually.1 Early detection of CRC can significantly improve patient outcomes and require less invasive treatment.2
The introduction of quantitative faecal immunochemical tests (FIT) marks a pivotal evolution in CRC screening. Older, qualitative guaiac-based tests (gFOBT) are prone to dietary interferences and offer a simple positive or negative result. Quantitative FIT tests, such as the QuikRead go iFOBT, have no dietary restrictions and the test provides a precise measurement of the concentration of human haemoglobin in a stool sample for risk stratification. The concentration of faecal haemoglobin (f-Hb) has been shown to correlate with the risk of advanced adenoma and colorectal cancer, both in screening and in symptomatic patients.3
FIT is the most widely used test for CRC screening worldwide4 and it is recommended for example by the Council of Europe and by the American College of Physicians for individuals between 50-74 years old.2,5
FIT in evaluation of symptomatic patients
Although CRC screening is important in early detection of CRC, majority of CRC cases are diagnosed after patients develop symptoms6. Diagnosing CRC in symptomatic patients is challenging because symptoms overlap with both common benign conditions and other serious bowel diseases, including advanced adenomas, ulcerative colitis, and Crohn’s disease.
In primary care, FIT can support triage by helping identify patients who are more likely to benefit from further investigations and prioritizing the urgency of referrals. Patients with FIT result below the cut-off whose symptoms persist or remain clinically concerning may be managed with appropriate safety netting or directed to further examinations depending on their symptoms and clinical presentation.7
How does quantitative FIT aid the diagnostic pathway?
One of the most powerful features of quantitative FIT tests is the ability to adjust the cut-off threshold. For instance, a screening programme can set a specific cut-off to balance sensitivity and specificity according to its specific needs and available colonoscopy capacity.4,8
In regions with limited endoscopy resources, the cut-off can be raised to prioritise patients at highest-risk, helping ensure that available colonoscopy capacity is directed to those who most need urgent investigation. In settings with more capacity, a lower threshold can be used to capture a wider net of potential cases. This adaptability makes quantitative FIT also a dynamic management tool, allowing health systems to adjust the flow of patients into secondary care based on real-time capacity.4,8
A simple, in-practice instrument, like the QuikRead go Plus, delivers accurate results directly at the clinic, allowing primary care professionals to make informed decisions about the urgency of a referral.
What is the evidence for cost-effectiveness of FIT testing?
A comprehensive Australian study found that biennial FIT screening is highly cost-effective, and when compared to screening with colonoscopy every 10 years, annual or biennial FIT has been shown to be either cost-saving or very cost-effective, particularly when high patient participation rates are achieved.9
In triaging symptomatic patients, The National Institute for Health and Care Excellence (NICE) in the UK recommends offering FIT in primary care to guide referrals for suspected CRC. This endorsement is built on strong evidence that FIT can safely rule out CRC in a significant proportion of patients, thereby streamlining the diagnostic process and making better use of limited endoscopic resources.10
FIT can provide cost savings through:
- Reduced procedural costs: Direct savings from avoiding unnecessary colonoscopies. FIT effectively rules out a large cohort of low-risk individuals, reducing the total number of expensive and resource-intensive colonoscopies.
- Lower treatment costs: Earlier detection of CRC leads to diagnosis at a more localised stage. Treatment for early-stage CRC is less invasive and less costly than treating advanced, metastatic disease, which often requires extensive surgery, chemotherapy, and long-term care.
- Optimised resource allocation: Endoscopy units can focus for patients at higher risk for CRC, enhancing the overall efficiency of hospital services. Staff and facility time is used more efficiently, making better use of existing resources.
How does QuikRead go iFOBT benefit in primary care?
Point‑of‑care (POC) FIT testing delivers results directly at the clinic. It eliminates the need to send samples to a laboratory, thus removing both transport and processing delays. In evaluation of symptomatic patients FIT enables faster decision-making and improved risk-stratification of the patients.
The QuikRead go iFOBT test, used together with the portable and fully automatic QuikRead go Plus or QuikRead go instrument, is easy to integrate into the primary care workflow. QuikRead go iFOBT offers:
- Informed decision-making: Accurate, instrument-read quantitative f-Hb result is delivered in two minutes. Adjustable cut-off values (10-200 µg/g in faeces or 50-1000 ng/ml in test solution) support precise risk assessment and help clarify the next steps.
- Better patient experience: The test is non-invasive, sample can be collected at home, and the 5-day sample stability provides flexibility. With the result, patient receives a clear assessment without prolonged uncertainty.
- Enhanced consultation quality: With objective data in hand, the GP can confidently discuss the next steps with the patient, either reducing anxiety from unnecessary referrals or reinforcing the importance of further investigation through a colonoscopy.
- Workflow integration: The test process requires minimal hands-on time and can be easily adopted into practice workflows. Samples are fully traceable via easy integration with LIS/HIS.
Three simple steps to the result
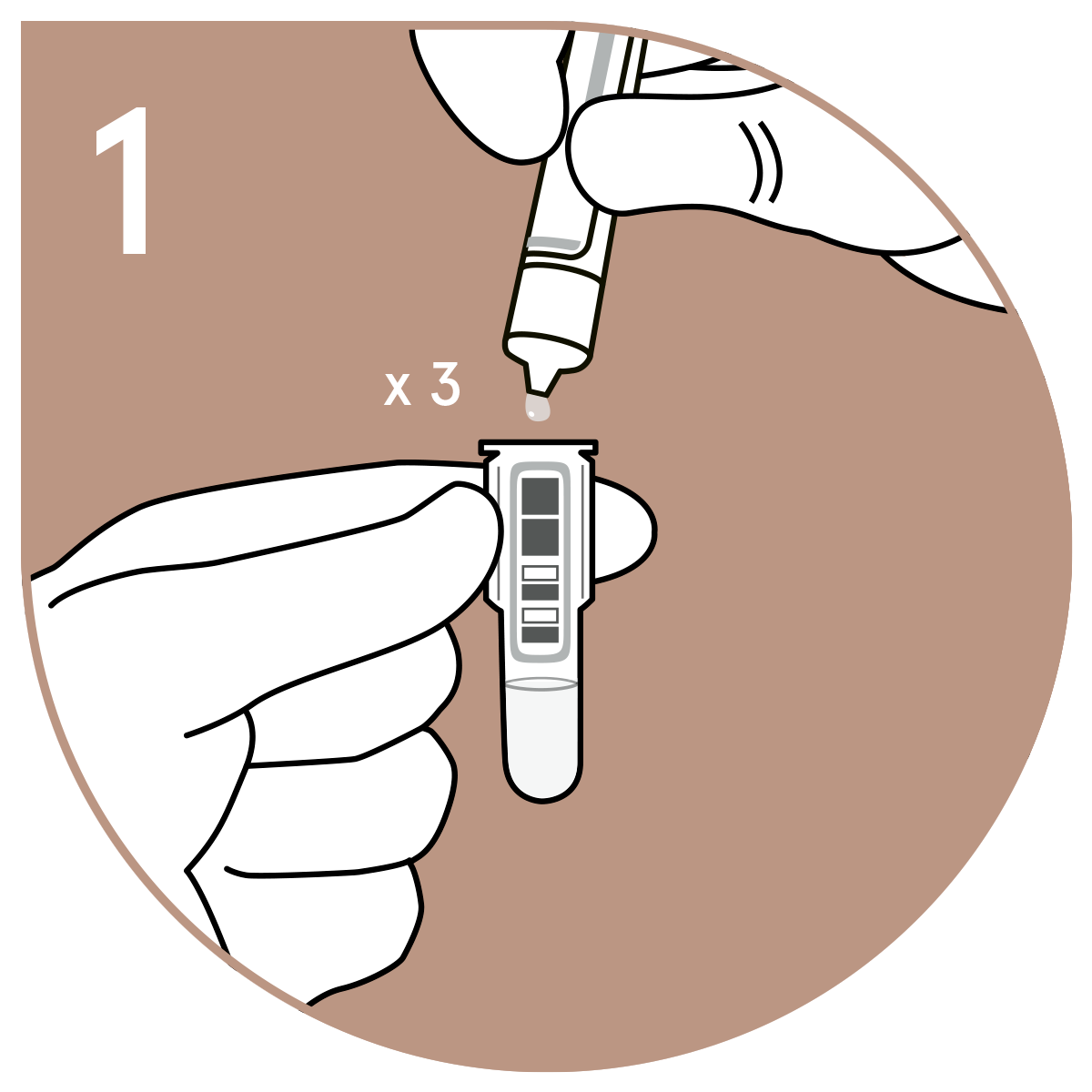 | 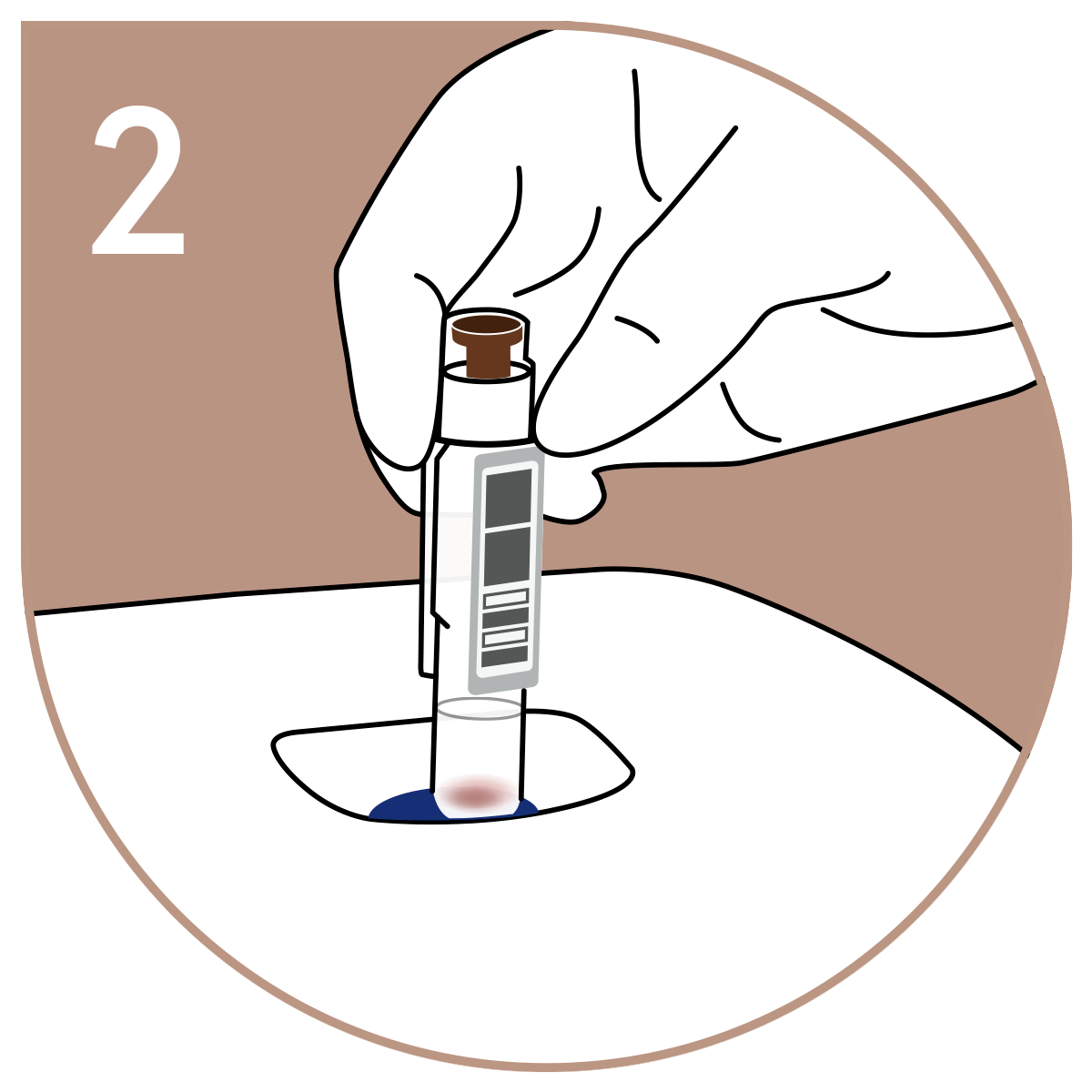 | 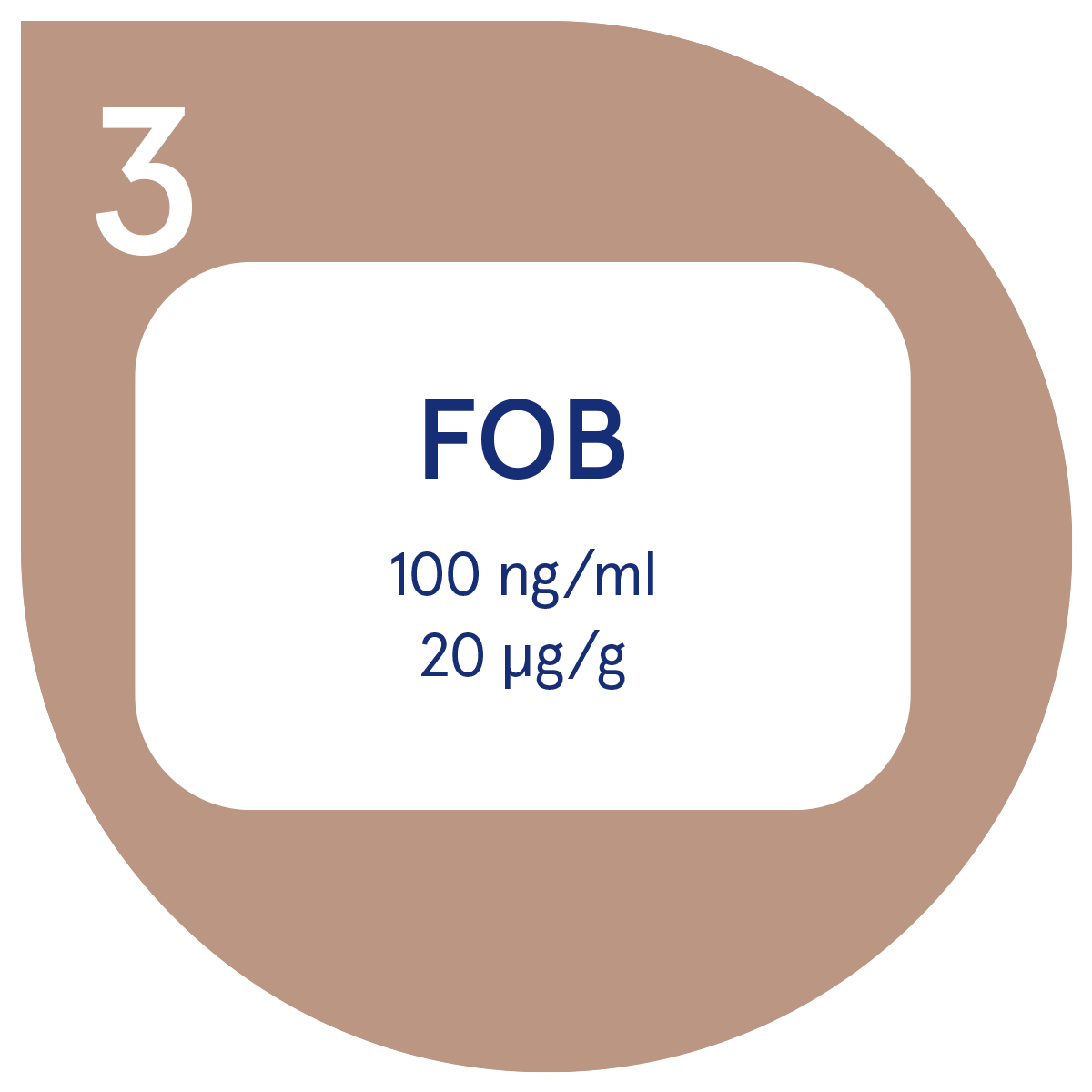 |
How does FIT optimise resources across the healthcare system?
The implementation of a quantitative FIT in CRC diagnostic pathway adds efficiency that benefits the patient, the GP practice and the hospital:
- For the patient: A faster, less invasive journey. A negative FIT result provides immediate peace of mind and spares patients the discomfort, inconvenience, and potential risks of an unnecessary colonoscopy. Those who do have a positive result can be referred to a colonoscopy for further examination.
- For the GP: A tool for confident triage and practice efficiency. Quantitative FIT provides an objective, evidence-based tool to manage symptomatic patients, reducing clinical uncertainty and unnecessary referrals. The improved quality of referrals helps improve cooperation between primary and secondary care.
- For the hospital: Freeing up capacity and maximising value. Hospital endoscopy units might be struggling with long waiting times and FIT can be used to prioritize the colonoscopy needs. Reducing the number of low-yield colonoscopies, FIT frees up capacity within endoscopy units for specialists to focus on patients with the highest clinical need.
Embracing a smarter standard of care
The future of effective and sustainable CRC diagnostics lies in optimising the entire patient pathway, starting in primary care. Quantitative FIT, as delivered by point-of-care systems like the QuikRead go Plus, is a great clinical and economic optimisation tool. It allows GPs to make quick, confident decisions, ensures patients are directed to the right level of care at the right time, and enables hospitals to allocate resources with maximum efficiency and impact.
Frequently asked questions
-
A FIT test (Faecal Immunochemical Test) is a non-invasive screening tool that detects hidden blood in stool samples by measuring human haemoglobin concentration.
Unlike older guaiac-based tests (gFOBT), FIT tests specifically target human haemoglobin, eliminating dietary interference and improving accuracy. It is widely used in primary care and national screening programmes to identify patients at risk of colorectal cancer who may require further investigation via colonoscopy.
-
Quantitative FIT result allows using a predetermined cut-off value (e.g. 10 µg Hb/g). A result above the cut-off is defined as a positive result. The majority of positive results are caused by other conditions such as adenomatous polyps, inflammatory bowel disease, haemorrhoids, or other benign sources of gastrointestinal bleeding. This is why quantitative FIT testing is valuable, it enables GPs to assess disease severity based on f-Hb concentration supporting more informed referral decisions.
-
Qualitative FIT provides a simple positive or negative result, while quantitative FIT provides the exact concentration of faecal haemoglobin (f-Hb) in µg Hb/g faeces. This distinction is clinically significant. Quantitative results enable evaluation of the disease severity and risk stratification as higher f-Hb levels correlate with greater likelihood of significant colorectal pathology. Healthcare systems can also adjust positivity cut-off thresholds based on colonoscopy capacity, making quantitative FIT a dynamic pathway management tool rather than just a screening test.
-
A quantitative FIT testing is recommended as a primary population screening test for CRC especially in Europe and other countries with stool-based national programmes. FIT is also increasingly used in symptomatic patients presenting primary care with lower gastrointestinal symptoms suggestive of colorectal cancer, such as pain, altered bowel habits, rectal bleeding, or unexplained weight loss. For example, in the UK, clinical guidelines recommend offering FIT to help triage patients and guide referral decisions for suspected CRC in primary care. Point-of-care tests like the QuikRead go iFOBT enable physicians to obtain quantitative results within minutes, allowing evaluation of disease severity, risk stratification, and pathway decisions during the consultation.
-
FITs have good diagnostic accuracy for detecting CRC in both screening and symptomatic patient pathways. It is particularly useful in identifying people who are more likely to benefit from further investigation, while it also aids in ruling out CRC in many low-risk patients.
Its performance depends on the cut-off used and the clinical setting. In general, a lower cut-off improves sensitivity, whereas a higher cut-off improves specificity. FIT is especially valued for its high negative predictive value, meaning that a negative result makes colorectal cancer less likely.
However, FIT does not detect all cancers or advanced adenomas, and results should always be interpreted together with symptoms and other clinical findings. Patients with persistent, concerning, or high-risk symptoms may still need further assessment even if the FIT result is negative.
Contact us for more information
References
- World Health Organization. Global Cancer Observatory database. https://gco.iarc.fr/ Accessed 3.3.2026.
- European commission. European guidelines for quality assurance in colorectal cancer screening and diagnosis – First Edition. Segnan N, Patrick J, von Karsa L (eds.), 2010, Luxembourg.
- Navarro M et al. Fecal hemoglobin concentration, a good predictor of risk of advanced colorectal neoplasia in symptomatic and asymptomatic Patients. Front Med (Lausanne) 2019; 6:31.
- Shaukat A & Levin TR. Current and future colorectal cancer screening strategies. Nat Rev Gastroenterol Hepatol. 2022; 19:521-531. https://doi.org/10.1038/s41575-022-00612-y
- Rex D et al. Colorectal Cancer Screening: Recommendations for Physicians and Patients from the U.S. Multi-Society Task Force on Colorectal Cancer.Am J Gastroenterol 2017; 112:1016-1030.
- Hamilton W & Bailey SER. Colorectal cancer in symptomatic patients: How to improve the diagnostic pathway. Best Practice & Research Clinical Gastroenterology 2023; 66:101842.
- Monahan KJ et al. Faecal immunochemical testing (FIT) in patients with signs or symptoms of suspected colorectal cancer (CRC): a joint guideline from the Association of Coloproctology of Great Britain and Ireland (ACPGBI) and the British Society of Gastroenterology (BSG). Gut 2022; 71(10): 1939–1962. https://doi.org/10.1136/gutjnl-2022-327985
- Pin-Vieito N et al. Optimal diagnostic accuracy of quantitative faecal immunochemical test positivity thresholds for colorectal cancer detection in primary health care: A community‐based cohort study. United European Gastroenterol J 2021; 9(2):256–267. https://doi.org/10.1177/2050640620949714
- Lew JB et al. Evaluation of the benefits, harms and cost-effectiveness of potential alternatives to iFOBT testing for colorectal cancer screening in Australia. International Journal of Cancer 2018 Jul 15;143(2):269-282. https://doi.org/10.1002/ijc.31314
- National Institute for Health and Care Excellence (NICE). Quantitative faecal immunochemical testing to guide colorectal cancer pathway referral in primary care (HTG690), 2023.


